Sodium ion batteries have been viewed as promising alternatives to lithium ion batteries for large-scale energy storage. But one of their problems has been a rapid degradation after a few charge/recharge cycles. One reason is that sodium ions (1.02 Å) are nearly twice as large as lithium ions (0.59 Å). The large size causes big changes in the host structure as the sodium ions come and go, causing a drop in capacity after repeated cycles.
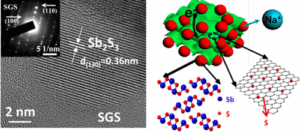 Researchers in China and Taiwan say they have come up with a new anode material that may help yield sodium-ion batteries that don’t degrade quickly. According to their paper published in ACS Nano, they bond nanostructured antimony on sulfur-doped graphene sheets (Sb2S3/SGS). The resulting batteries are relatively stable, retaining 83% of their capacity for 900 cycles. (For comparison, Apple says the iPhone battery is designed to retain up to 80% of its original capacity at 500 full charge and discharge cycles, while a MacBook Pro is designed to deliver up to 1,000 charge/discharge cycles before dropping to 80% of its original capacity.)
Researchers in China and Taiwan say they have come up with a new anode material that may help yield sodium-ion batteries that don’t degrade quickly. According to their paper published in ACS Nano, they bond nanostructured antimony on sulfur-doped graphene sheets (Sb2S3/SGS). The resulting batteries are relatively stable, retaining 83% of their capacity for 900 cycles. (For comparison, Apple says the iPhone battery is designed to retain up to 80% of its original capacity at 500 full charge and discharge cycles, while a MacBook Pro is designed to deliver up to 1,000 charge/discharge cycles before dropping to 80% of its original capacity.)
Researchers say the cycling performance of the Sb2S3/SGS composite exceeds that of any other antimony-based materials. They also say computational calculations show that sulfur-doped graphene (SGS) has a stronger affinity for Sb2S3 and the discharge products than pure graphene, resulting in a robust composite architecture for outstanding cycling stability.
Of course, there is still a long road ahead before commercialization of the technology becomes feasible. To commercialize the idea, researchers would need to scale up battery fabrication while maintaining its high performance.


Leave a Reply