It pays to know how a testing regime can reveal whether specific types of lithium batteries will last as long as an application demands.
SOL JACOBS, TADIRAN BATTERIES
 DESIGNERS MUST UNDERSTAND how their end device will consume power before they can choose a battery. Will the battery be depleted in a matter of hours, as for an army manpack communications radio? Will the device spend most of its time in sleep mode and transmit data bursts several times daily, as is the case with most wireless sensors? Does the device require a 10+ year operating life?
DESIGNERS MUST UNDERSTAND how their end device will consume power before they can choose a battery. Will the battery be depleted in a matter of hours, as for an army manpack communications radio? Will the device spend most of its time in sleep mode and transmit data bursts several times daily, as is the case with most wireless sensors? Does the device require a 10+ year operating life?
Answers to these and other key questions will help determine the ideal power supply. You may want to run your own tests — engineers at Tadiran can assist you in establishing the proper test procedures. Make sure you run ‘apples to apples’ comparison tests using similar types of batteries, as comparing totally different types of batteries could yield highly inaccurate results. For example, it is unrealistic to compare cells designed to supply moderate rates of current with those designed to supply low average daily rates of current.
Among primary lithium batteries, there is a natural trade-off between two kinds of cells: those designed to work for the long-term with a low usage rate and low-self-discharge; and those offering short-term operation with a higher usage rate but higher self-discharge.
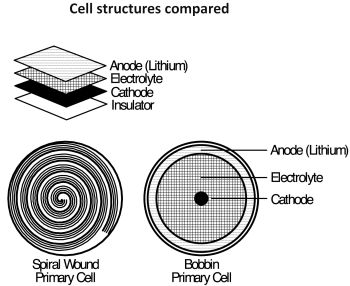
Certain lithium batteries, including lithium thionyl chloride (LiSOCl2) chemistry with spirally wound construction, lithium sulfur dioxide, and lithium manganese dioxide, can deliver energy at a high rate for relatively brief periods. Spirally wound LiSOCl2 cells have electrodes with large surface areas which support higher rates of output. However, the downside of spiral-wound construction is a significantly higher self-discharge rate. By contrast, bobbin-type LiSOCl2 cells feature much less electrode surface area, supporting a lower rate of output along with a lower rate of self-discharge.
A simple analogy between these two types of batteries would be an 8-oz. (capacity) glass of water. Over time, the large opening of the glass allows the water to evaporate faster (higher self-discharge). But the larger opening allows the water to pour out rapidly (higher rate of output). The wider opening serves up energy immediately in applications thirsty for large amounts of energy. However, if you want to keep that water around for an extended period of time, the wider opening becomes problematic, allowing the water to evaporate more quickly.
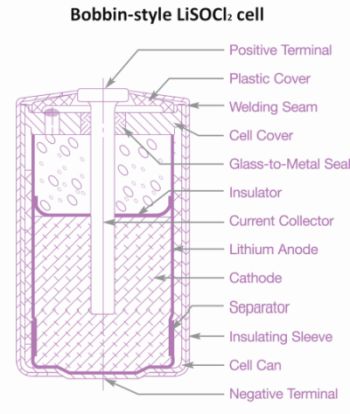
Certain bobbin-type LiSOCl2 cells, including the Tadiran 59XX IXTRA Series, were designed to supply a moderate rate of discharge over a relatively long operating life with a low annual self-discharge rate ranging from 1 to 2% a year. In storage, these batteries would use up approximately 10-20% of their initial capacity over ten years. This performance has analogies to a soda can with a moderate opening — the smaller opening lets the soda evaporate more slowly than from a large glass with the trade-off that the liquid can’t pour out as fast.
Tadiran manufactures the XOL EXtended Operating Life cells (49XX family), which features the lowest self-discharge rate of any lithium battery commercially available, which allows you to draw a low rate of current while featuring a low rate of annual self-discharge, just 0.7% per year. Think of it as a bottle of water with a small opening and a straw that allows only a sip of water, while limiting evaporation to a slow rate.
To create a bobbin-type LiSOCl2 battery able to deliver high current pulses while offering a low rate of annual self-discharge, Tadiran developed PulsesPlus batteries. They contain a patented hybrid layer capacitor (HLC) to store and release high pulses of energy without a higher rate of self-discharge. The analogy is a plastic bottle: Squeeze it when you want to squirt the water (high pulse). When you are not squeezing the self-discharge is still extremely low, similar to the XOL cell.
With any battery that uses a capacitor, the capacitor should never get fully exhausted of energy while supplying the pulse. This has much to do with the capacity of the capacitor in Farads. If capacitor charge capacity is depleted during the generation of the pulse, the battery might have to supply some of the pulse energy. In cold temperatures or toward the end of the battery’s useful life, the resulting current drain could cause the battery voltage to drop too low and cause the device to
shut-off.
HLCs manufactured by Tadiran have significant energy storage capacity starting at about 100 F. With so much capacity, there is little chance the battery will ever be called upon to directly supply more than the low current necessary to charge the HLC. By contrast, standard capacitors can have a much smaller capacity ranging from a few hundred millifarads to about 10 F.
Comparative testing
Consider our analogy again: It takes longer to pour 8 oz. of water through a small straw than from a glass. Similarly, it takes much longer to run a test that shows how much water is in the can than one that shows the amount in the glass. You also need to make sure that you are comparing straws to straws and cans to cans. Otherwise it will look like the cans and glasses have more capacity than the bottles, which is not true. Also, how do you properly measure the evaporation (self-discharge)?
Needless to say, properly testing a battery is time-consuming and intensive process that demands appropriate test methods to yield valid results.
Tadiran has been designing and manufacturing lithium primary batteries for over 30 years. The company tests its batteries under various conditions, covering almost every possible scenario. In addition, it collects data based on factors such as cell size, temperature, load size, and so forth, under different loads. This knowledge has been organized into a comprehensive matrix of battery status information covering many different application requirements.
When recommending a battery for use in a specific application, Tadiran draws upon a vast library of historical test points, including OC (over charge) voltage, voltage under load, annual self-discharge rate, low-voltage point at load, and temperature. This comprehensive data matrix is then used to calculate long-term dissipation of available cell capacity based on application-specific requirements. These calculations are based on actual long-term test data, not theoretical projections.
Here is a brief synopsis of the various types of testing methods that Tadiran employs:
Actual long-term testing – Tadiran’s lab has batteries still being tested after 25+ years and which are still operating. These batteries are being tested under different loads and
test profiles.
Accelerated testing – using a method called Arrhenius test (two-fold increase of reaction rate for every 10°C) we can reduce the amount of time needed to simulate long-term operation. These tests take place at 72°C, which is the equivalent of about 32 times the theoretical lifetime of a battery at 22°C. It is important to properly run and interpret results from this test, as an inferior cell that suffers from passivation could indicate a false positive result. For this reason, it is essential not only to store the cell at 72°C prior to testing, but to actually test the cell during storage at 72°C. Here is one example of this type of testing:
Some cells are tested at 72°C for one month at the one-month discharge rate (depleting the cells). Others are tested for two months at the two-month discharge rate. Still others are tested for 3-,4-,5-, 6-month and one-year discharge rates. At the one- and two-month discharge rates, Tadiran XOL cells have demonstrated low capacities because they are not designed to discharge quickly. (Recall a lithium battery can have a high usage rate with high self-discharge or low usage rate with low self-discharge rate, but NOT both). By contrast, Tadiran IXTRA cells, as well as competitive products, will show higher capacities at fast discharge rates. But these tests also indicate that their self- discharge rate will also be higher.
Starting with month three, testing at the three-month discharge rate at 72°C (the equivalent of eight years at 22°C) will show competing cells to have higher self-discharge rates, indicating that Tadiran cells can deliver a longer operating life thanks to lower self-discharge. The longer the test proceeds with a low usage rate, the better Tadiran cells will perform because of their lower self-discharge. To confirm this, Tadiran has run this test on cells for 90 months at 72°C (the equivalent of hundreds of years of continuous operation).
Calorimeter testing: A battery is surrounded by water and then calorimeters are used to look for a rise in temperature caused by self-discharge. This test runs only after a battery has completely stabilized for a period of one year prior to being tested.
Field results: Laboratory test results continue to be proven and confirmed by actual results from the field. Tadiran often checks random samples of batteries in long-term use to see if the amount of lithium left in the battery coincides with the results from the lab. Another valuable metric is the number of Failures In Time (FITs) measured in billions of device operating hours in the field. Tadiran’s historical average FIT is between five and 20 batteries per billion, extremely low compared to the industry average.
Lithium Titration: There are specialized applications with unusual requirements where sufficient data points may not be available (temperature extremes, prolonged high current pulses, short life time applications etc.). In these cases, Tadiran measures the remaining content of the lithium anode in the battery after specific test conditions (e.g. partial discharge, temperature soaking and
so on).
An example might be after a battery has been tested for several months under elevated temperature and various discharge currents. It is then cut open to dissolve the remaining lithium. Results from the titration can predict annual battery self-discharge as a function of the applied currents and/or temperature, because the higher the self-discharge rate, the less lithium remaining in the cell.
Competitive Testing: Tadiran routinely runs lab tests on competing batteries. But first, we typically operate the battery for one year, which allows any impurities in the electrolyte to show up in the test results.





Leave a Reply