Engineers trying to shield RF-sensitive circuitry a few years from now may have the option of using material that absorbs RF energy rather than just reflecting it.
So say researchers at Drexel University and the Korea Institute of Science and Technology. They are researching a class of material called MXenes, and more specifically, titanium carbonitride MXene that can be processed in a way that lets it absorb electromagnetic radiation.
First described in 2011, MXenes are called two-dimensional compounds because they have a structure consisting of layers that are only a few atoms thick. The material itself consists of transition metal carbides, nitrides, or carbonitrides. It exhibits a high conductivity because of the metal content and is hydrophilic because the surfaces comprising it have hydroxyl or oxygen terminations.

Researchers stumbled onto the RF absorption property when they annealed titanium carbonitride MXene at 350ºC. The heat oxidized the material enough to remove intercalated water and hydroxyl surface terminations and thereby push the surfaces closer together as well as give them some porosity.
For reasons that are not yet clear, the resulting material was able to absorb RF. “The mechanism is still not fully understood,” says Drexel College of Engineering Professor Yury Gogotsi, leader of the research group. “It’s certainly different from bulk metal. In the microwave frequencies range, the dependence if fairly flat. However, in the visible and IR range there are pronounced peaks in the absorption spectrum due to surface plasmons or electronic transitions. We need to systematically cover the entire electromagnetic spectrum to better understand absorption properties of MXenes. Keep in mind that we have dozens of different MXenes already produced and their properties vary widely, sometimes in unexpected ways.”
Researchers characterized the RF absorption behavior at 8.5 GHz in work covered by an article for a recent issue of Science Magazine. Gogotsi says the team hasn’t exhaustively measured the bandwidth over which the RF absorption takes place, but “We expect it to be very broad. We have previously shown very efficient absorption in the terahertz range,” he says.
As you might expect, absorbed RF energy gets converted to heat in the MXene material. “We didn’t quantify the heat effect, but the heating of MXenes by RF waves has been previously reported. I expect the amount of heat released in the case of telecommunication or radar stealth applications is negligible, but certainly careful measurements are needed,” Gogotsi says.

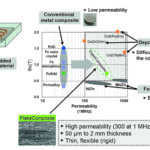
40-mm-thick Ti3CNTx and Ti3C2Tx films in
the X-band frequency range after annealing
at various temperatures.
It also looks as though production of RF-absorbing MXenes could be scaled up to commercial quantities using the same basic process employed for research work. “We start with a layered ceramic known as MAX phase, e,g., Ti3AlC2 or Ti3AlCN in this particular case, and etch away monoatomic layers of aluminum in acid to release nanometer-thin layers of titanium carbide or carbonitride,” Gogotsi explains. “Thus, we cannot start with bulk cubic titanium carbide or carbonitride. However, the MAX phases are produced in a ceramic synthesis process similar to synthesis of TiC or TiCN powders, so we have a comparably scalable precursor. We can produce 50-100 g of MXene in a single batch in our lab, so this will translate to enormous area of the EMI shielding film. For perspective, consider that 40-50-nm films of Ti3C2 can absorb 99%, and one-micron films 99.999%, of EM radiation in the X-band.”
The Drexel team has examined EMI absorption capabilities of 16 different MXene materials, about half of all those produced in its lab. And the promise of a more sustainable way of handling EM pollution has attracted the attention of industry. “The technology has been licensed from Drexel to a Fortune 1000 electronics corporation which is moving towards commercialization of MXene-based EMI shielding materials,” Gogotsi says. “And we’ll have a panel on MXene commercialization at our MXene conference in August. More than 1,500 people from about 60 countries registered. The interest to MXenes keeps growing.”




Leave a Reply